Page Contents
We’ll start with an overview of how to image a fluid flow phenomenon. ‘Fluid flow phenomenon’ can be anything that gases or liquids or even plasmas can do. We’ll use the ‘how’ to structure the information, and sprinkle in the basics of photography, physics of fluid flows, optics and light/matter interactions as we go along. After this initial overview, we’ll focus atmospheric clouds as an example of how flow visualization (hereafter shortened to flow vis) reveals the physics of the sky. Then we’ll go into specific flow vis techniques in more detail.
The Choices
Before you begin, it’s best to have an idea of what you are trying to do. Sure, sometimes you’ll just stumble across a fantastic flow and capture a great image, but here we are talking about a more thoughtful type of work, of artwork. A definition of art that I believe in is that art is the instantiation of a vision: having an idea and making it so. Definitions of art and science can get deep, so we’ll address them later. For now, try to previsualize: have a goal, i.e. think about what you want it to look like.
We can frame this process as a series of choices:
- Flow phenomenon: Water boiling? Faucet dripping?
- Visualization technique: Add dye? See light distorted by air/water surface?
- Lighting: Continuous? Strobe? Sheet?
- Visual acquisition: Still? Video? Stereo? Time lapse? High speed?
- Post processing: creating the final output. Editing, at least cropping the image and setting contrast.
1. Flow Phenomenon: Why Does It Look Like That?
Your inspiration for phenomena can come from any aspect of your life: your work as a scientist, engineer or artist; your kitchen or bathroom; some flow you saw online; or the natural world outdoors. But in order to make an image that walks the line – that satisfies both art and science and is thus richer than either alone – ask as you work with the fluid, “Why does it look like that?” Seeking the answer to this question throughout the process of creating your image will give you clues to control the flow to make it match your vision, or possibly inspire you to change your vision. It can be an iterative process. You may be inspired to “work it,” to make variations and explore new aspects. Answering that question can also give deeper meaning to your image.
It’s a big question, though, and hard to approach, especially for those new to spending quality time with fluid flows. Considering what forces may be acting on the fluid is often a useful starting point.
Minute Paper: list all the forces that can act on a fluid
Take a few minutes to think for yourself before reading these answers from class.
Viscous
Shear
Gravitational
Buoyancy
Electromagnetic
Electrostatic
Inertial
Centripedal/centrifugal
Pressure
Body forces: gravity, buoyancy, EM
Viscosity, shear, friction
Thermal diffusivity
Interaction with other fluids
Surface tension
Intermolecular
Air resistance (drag)
Cohesion
Adhesion (capillary action)
Normal force
Stress
Strain
Thermodynamic
Heat
Convection
Osmosis
Solar radiation
Composition of fluids
Densities of fluids
Chemical reactions
Impact
Wind
Mass
Acceleration
Temperature
Phase change
Strong, weak nuclear forces
Cavitation
Vortex structures
Vortex stretching
concentration gradient
This is a good, inclusive list. Although not all of these items are forces, all can ‘drive’ a flow via some mechanism or set of physics. Heat, for example, is not a force per se, but can certainly induce a flow to move by reducing the local density of a fluid so that neighboring high-density fluid will push it upwards. It’s actually pressure that makes the low-density fluid move upwards, since the pressure in a static fluid is directly related to the density in the column above it.
In mechanics, a force is specifically a push or pull . Forces can be categorized into two types: body and surface.
Body Force
This type of force acts on every molecule equally. Examples include gravity and magnetism.
Surface Force
Consider an imaginary cube of a fluid, suspended in an identical fluid. Pressure is the force per unit area acting perpendicular to each surface of the cube (a.k.a the normal stress), and is due to neighboring molecules banging up against the (imaginary) surface. Shear is the force acting along the surface, parallel to it. You can think of it like friction. It’s also due to the neighboring molecules.
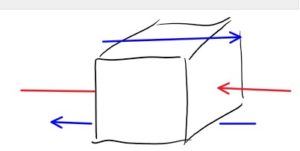
Any force acting on the surface of the cube can be decomposed into a pressure plus a shear.
Surface Tension

Surface tension is the result of molecules being attracted to other molecules in the fluid, but not to molecules in the gas, above the surface. Roland.chem, Public domain, via Wikimedia Commons
It’s the result of intermolecular forces, so it affects every molecule, like a body force, but it is only obvious at interfaces between liquids, or between liquids and gases, kind of like a surface force. Molecules that are fully inside the liquid are equally attracted to all of its neighbors; this is ‘cohesion’. Molecules at the surface are attracted to those at the side and below, but not to the gas above. (There aren’t enough molecules up there to do much anyways.) The net result is that the surface behaves as though it is under tension; hence, “surface tension.” If the chemical composition in one area of the surface is different from another area or if the temperature is different, they will have different surface tensions, and fluid will be dragged from the low-surface-tension area into the higher. This effect is called ‘Marangoni convection’ , and is responsible for all sorts of interesting visuals.
Figure 3: A droplet of Axe golden glitter shampoo creates a region of low surface tension in dyed milk, resulting in Marangoni flow. By Alex Meyer, Dillon Thorse, and Jeff Pilkington, Team Second, Spring 2013.
For example, Figure 3 shows that a surfactant – here, a drop of shampoo – lowers local surface tension, allowing the surrounding fluids (milk and food dye) to pull fluid away from the drop.
Choice 1 Conclusion: whenever you are observing fluids, list the forces that may be acting, that make it look like that.

