Why are there clouds? After all, clouds are made of tiny droplets of water or ice particles, right? Both are much heavier than air, so why don’t they just fall? Here’s why: the air around the droplets is moving upwards faster than the droplets are falling! There are no clouds without atmospheric lift! You can understand a lot about clouds and weather if you get that one simple fact.
Even better, the reason for that atmospheric lift, the lift mechanism, determines appearance of the cloud, so knowing the lift mechanism you can figure out the cloud, and vice versa to some extent. Here are the three major lift mechanisms:
- Instability: a less dense pocket of air surrounded by denser air will rise. Notice how it’s all about density, mass per unit volume, and not just about temperature. Stable air leads to stratus clouds and unstable air leads to cumulus.
- Orographics: air gets pushed up by the terrain, especially mountains. The mountains act as a trigger for a variety of cloud types.
- Convergence: weather systems, at both warm and cold fronts can lift a mass of air as a whole over hundreds of square miles. Depending on location within a typical system you could get any of the 10 genera.
Instability
This mechanism is responsible for all of the cumulus cloud types. Here in Colorado, instability accounts for almost all the clouds of summer, and about 1/4 of winter clouds. It is also the most complex mechanism to wrap your head around, and requires looking at the basic thermophysics of the atmosphere. Oooh, that sounds scary even if you’ve had a class in thermodynamics, but don’t worry, we’ll stroll through it at a comfortable pace, and you’ll find a lot of it is intuitive for all of us walking bags of water, swimming in our ocean of air. We all live with this stuff every day; this is just a specific way of looking at it.
An instability is something just waiting to happen, like a ball on top of a hill, or a helium balloon held in a child’s fist. The slightest trigger will set it off; nudging the ball, or the kid letting go of the string. Clouds result from the Rayleigh-Taylor instability , with denser air sinking through a less dense region and vice versa. A familiar human-scale example is water and oil; if the water (more dense) starts on top of the oil (less dense), it will fall through the oil. Water on top is an unstable situation, whereas water on the bottom is stable. In the case of cumulus clouds, a parcel of air is warmed by contact with the ground — maybe a hot parking lot — and becomes less dense than the neighboring parcels. It rises, and continually finds itself less dense than its neighbors, so it keeps going up. It cools as it goes (we’ll see why shortly) until the water vapor in the parcel is cool enough to condense into a cloud. What happens next… depends.
The situation in the atmosphere is more complex because we are dealing with scales large enough that the air near the ground and air high enough to form clouds are at different pressures, temperatures and densities. Plus the properties of air-water mixtures depend on whether the water is liquid, vapor, or changing between. Before we proceed, we need to see how pressure, density and temperature vary normally in a column of atmosphere.
Layers of the Atmosphere
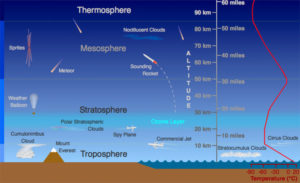
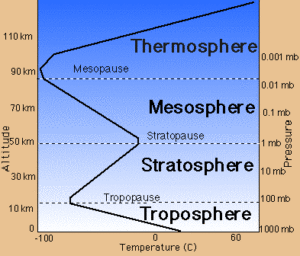
Figure 1 shows how the layers are approximately defined by temperature. Starting at the ground, temperature decreases as we go up through the troposphere. Almost all clouds and weather happen in the troposphere, with just a few rare clouds above it. Then comes the tropopause, a thin layer where the temperature stays constant. Then we go into the stratosphere where temperature increases. Figure 2 shows this in more detail, and also shows the pressure decreasing as altitude increases.
Density is a function of both pressure and temperature, and these are changing in different ways as we go up through the troposphere and stratosphere. We need to know more before we can say much about stability in the atmosphere.
The ozone layer is in the stratosphere. Ozone absorbs ultraviolet light from sunlight, heating the stratosphere .
Hm, Figure 2 is all in SI units. If you grew up here in the US, you might not have an intuitive grasp of how big 10 km is. In talking about the atmosphere, it’s helpful to be able to guesstimate these large length scales in both SI and English units, at least to one significant figure.
Try this now!
See if you can guess these off the top of your head, approximately:
- How big is one mile in km?
- How big is 10 km in miles?
- How many thousand feet in one km?
- How many km in 10,000 feet?
Now go ahead and google the answers.
I’ve memorized and forgotten all these values many times, so now I just remember a couple, and do the arithmetic from there: one mile is about 5000 feet, and one km is 0.6 miles. For today, keep in mind that the troposphere is 10 km thick, so that’s 30,000 feet, or 6 miles. You might remember that because commercial airliners fly at around that altitude, in the tropopause and lower stratosphere. That’s above almost all weather, and the air is less dense, so the planes don’t have to fight as much drag. For comparison, Longs Peak here in Colorado is a bit over 14,000 feet tall, so that’s almost halfway through the troposphere.
Adiabatic Lapse Rate
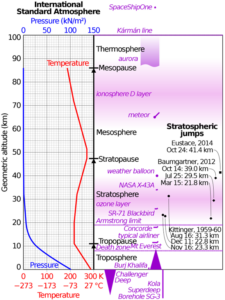
As Figure 2 shows (and you might have experienced), it’s colder up on top of Longs Peak. You can expect a roughly 5 degree F drop for every 1000 feet of elevation gain . But why? I’ll give you a hint: it’s not the Ideal Gas Law.
Let’s start with the pressure profile in a column of atmosphere. The air at the bottom is pressurized by all the air sitting above it. This is basic hydrostatics: gravity acting on the air is balanced by the pressure in the air at every level. Since it’s in balance, the air doesn’t move (hydrostatics). It’s almost a linear relationship in the troposphere, as shown in Figure 3. By the way, don’t worry about the units of pressure; they’re explained on the next page.
Now for the temperature part. Consider a parcel of air sitting in front of you: a little imaginary one inch cube holding a certain number of air molecules. The cube can change shape, but the molecules stay trapped inside, i.e., a ‘control mass’ for those of you who’ve learned some thermodynamics. The cube is here with you near the bottom of an air column, so it’s under one atmosphere’s worth of pressure, acting on all sides. Technically, the pressure on the top side is a tiny bit less than on the bottom, but the difference is small enough to ignore for a one inch cube. Now, let’s reduce the pressure from the surroundings. The cube will expand, yes? If we don’t allow any heat transfer — no conduction of heat or radiation through our imaginary cube walls — then the expansion is called adiabatic. By expanding, the cube is doing work on its neighbors; it expanded because we reduced the pressure outside, so the higher inside pressure pushed out over the distance of the expansion. A force acting over a distance is a definition of work. Since the air inside did work, it had to lose energy — specifically, internal energy. The molecules slowed down slightly, and we see this as a drop in temperature. It cooled due to the conservation of energy, the First Law of Thermodynamics. Not the Ideal Gas Law. Figure 4 is a pretty good simulation; check out the speed of the particles bouncing inside. If I knew the author I’d give credit.
Figure 4: Gas molecules in a cylinder compressed by a piston heat up and speed up as work is done on them. When allowed to expand, they do work and cool. Physics-animations.com.
Back to the atmosphere. If we take our little air parcel and raise it adiabatically by a few hundred feet or more — far enough that the local pressure really is significantly lower — our parcel will expand, doing work and cooling along the way. The reverse is true, too. If we take a parcel from a significant height and bring it down to ground level, it will be compressed; work will be done on it, and it will transform that work into heat. So if wind blows air up a mountain, it cools as it goes, and if wind brings it down, it will warm up. This mechanism is called the adiabatic lapse rate , and it mostly explains why the air on top of a mountain is cooler. But of course the reality of the atmosphere’s temperature profile is more complicated.
Air doesn’t simply move up or down adiabatically; there is always some heat transfer happening. How much water vapor is in the air plays a role, too. Water can store energy without changing temperature as much as dry air, so if there is a lot of water the moist air won’t cool as quickly with altitude. In addition, if the water condenses and forms a cloud, it releases heat and warms the adjacent air. Then the air/cloud mixture cools more slowly than dry air as it rises; this rate is called the moist adiabatic lapse rate. Most importantly, weather systems moving through mix up the atmosphere, bringing warmer or cooler air to various altitudes, as well as changing the amount of water vapor. As a result, the actual temperature profile can deviate a lot from the dry adiabatic lapse rate.
Now, let’s go back to the idea of instability. Imagine a bit of air heated by contact with a hot parking lot. It’s basically at the same pressure as the cooler air outside the parking lot, and this time the Ideal Gas Law says, yes, the density is lower. The parcel of hot, low density air rises and cools, pretty much adiabatically. A few hundred feet up, however, the neighboring air parcels may be more or less dense than our parcel, depending on the actual temperature profile in the air there at that moment. If our parcel is more dense than the neighbors, it will sink back down: the atmosphere is stable. But if the parcel turns out to be less dense than the neighbors, it will keep rising: the atmosphere is unstable. It only took a little kick from a hot parking lot to trigger a continuously rising column of air. If the atmosphere is unstable, a hot parking lot or a hot cityscape or a dark dirt field can trigger a column of air to rise high enough and cool enough to condense into a cumulus cloud, or even to make a thunderstorm. But to know whether the atmosphere is stable or unstable, we must measure the actual local temperature profile and compare it to the adiabatic lapse rate. That information comes from Skew-T data, described on the next page.
References

