Page Contents
Sometimes we don’t have to provide light because the fluid itself emits light. The most common example is a flame. Let’s start with a candle flame; how it is structured, and then how it emits light. After that we’ll describe other ways in which fluids can emit light.
The actor Alan Alda is interested in improving science communication. In 2012 he sponsored a contest that challenged scientists to answer the question “what is a flame?” in terms that an 11-year old could understand. He’d had this question himself ever since he was 11. The winner was a 7.5 minute animated video by a physics graduate student, Benjamin J. Ames, that has had over 700,000 views between Vimeo and YouTube.

Here’s a short version of what happens in a candle flame, illustrated in Figure 1. Heat from the flame liquefies the candle wax, which is then drawn up the wick by capillary action. The heat then vaporizes the wax, making a rich fuel vapor cloud which diffuses outwards. At the same time, air, including oxygen, diffuses inward, into the fuel cloud. When the oxygen mixes with the fuel at just the right concentration (stoichiometry), the combustion takes place. This is a chemical reaction that turns the air + fuel mixture into products: C02 + water vapor + a few nitrogen compounds (from the air) plus heat, heating up the fuel, air and product gases. During the combustion process, the wax molecules break apart and temporarily form excited CH* and C2* molecules. These particular molecules give off lovely blue photons during their short lives in the reaction zone. This is the blue glow you see at the bottom of the candle flame (see section 3.3 below). Meanwhile, the fuel vapor cloud near the wick gets heated by the reaction zone and starts rising due to buoyancy. The hot fuel vapor molecules start breaking apart, releasing carbon atoms, some of which start clumping together into soot particles. These soot particles are further heated by the nearby reaction zone and quickly get hot enough to glow bright yellow hot and swamp out the blue reaction region light. Most of them get burned up higher in the flame.

There are two main types of combustion: diffusion flames and premixed flames. The candle is an example of a diffusion flame. Not all diffusion flames make soot and glow yellow: isopropyl alcohol burns cleanly, never making soot. Rubbing alcohol contains water in addition to isopropyl, which cools the flame to a certain degree, but the flames are still hot enough to burn skin. Certain chemicals can be added to flames to make other colors, and the ‘rainbow flame experiment’ is a popular chemistry demonstration, but is surprisingly dangerous if not conducted properly. A number of students have been burned by their teachers during this experiment . In addition, the salts used to generate the colors make product gases that are toxic, so this is not a recommended experiment without extensive preparation and supervision. Other examples of diffusion flames are campfires, pool flames (Figure 2) and tiki torches.

The other type of flame, premixed, is very common in daily life. Natural gas flames in the home (cooktops, grills and water heaters) are premixed, as is the combustion in Bunsen burners, butane and acetylene torches (Figure 3), and gasoline-powered engines. ‘Premixed’ means the gaseous fuel and air are mixed somewhere upstream in the flow, before the mixture encounters the flame. The fuel/air mixture is usually controlled to be ‘lean’; have more air than the fuel needs to burn completely. So premixed flames generally don’t have a fuel-rich region and don’t make soot, and only emit the blue reaction-region light . Figure 4 shows a beautiful cellular flame: a small amount of rubbing alcohol is sloshed around in a large jug, filling the volume with premixed fuel and air. When a lighted match is dropped in the flame propagates from one end to the other, may push flame out the top (keep it open!) and periodically re-ignite. In general, as flows become faster and more turbulent, the line between diffusion flames and premixed flames becomes blurred, and partially premixed flames result. Fuel spray flames are also complicated, with regions of both premixed and diffusion-limited combustion.

Remember, if you are going to attempt a combustion experiment for the Flow Vis course, you must adhere to the combustion safety guidelines.
Maxmath12, CC0, via Wikimedia Commons
Incandescence
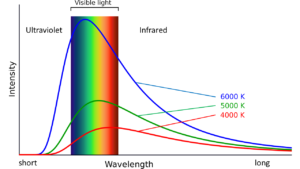
That yellow glow in the candle flame is from black body radiation, which you might consider a misleading name, since the soot is obviously glowing yellow. The name comes from the idea that black bodies both absorb and emit photons with a wide range of photon energies, i.e. colors. (If you’re not sure how photon energy is related to wavelength and color, check out this page). A lot of the photons will have a particular color that depends only on the temperature of the black body, as shown by the peaks in Figure 5. An object at 3000 K (6740 oF) peaks in the red, but glows with a warm white since it actually gives off photons with a wide range of energies, and therefore colors and wavelengths. A hotter object, say 5000 K, would look more blue-white and counterintuitively produce a ‘cooler’ white, and emit less, relatively, in the ultraviolet and infrared wavelengths because the peak is sharper. For comparison, the sun’s temperature is almost 6000 K .
Black body radiation is also called thermal emission of photons, or incandescence. In an incandescent or ‘Edison’ light bulb a tungsten filament is heated to incandescence. Figure 5 shows why incandescent bulbs are so inefficient; a large fraction of the photon energies are outside the visible light band. There is a clever quantitative flow visualization technique that uses a high-powered pulsed laser sheet to briefly heat soot or other particles to incandescence: laser-induced incandescence (LII) .
Photoluminescence = Fluorescence and Phosphorescence
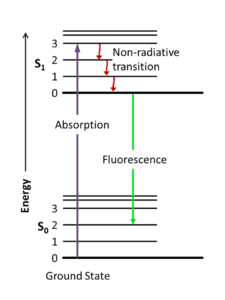
Photoluminescence happens when a fluorophore (e.g. a molecule or atom that can do this, can fluoresce) absorbs a photon at a specific short wavelength and emits a photon at a lower energy with longer wavelength. The absorption kicks the molecule from a ground electronic state into a higher energy state as shown in Figure 6. The difference in energy between the absorbed and emitted wavelength is called the Stokes shift, and may be due to heat lost from the excited state (non-radiative loss, so dropping from S1:3 to S1:0 perhaps) and/or the emission returns the molecule to a slightly higher ground state, such as S0:2 instead of S0:0. In fluorescence, the outgoing photon is emitted very quickly, while in phosphorescence there can be a lengthy time delay resulting in ‘glow-in-the-dark’ materials. The difference has to do with electron spin multiplicity in the materials ; let’s not get into that.

An example of fluorescence that you’ve probably seen but maybe not noticed is a fluorophore that is commonly added to laundry detergent and fabric softeners. High energy UV photons are absorbed from sunlight or a UV lamp, and emit blue or orange photons. Makes your clothes glow in sunshine! Whites look whiter, colors look brighter! Try shining a UV light on some detergent and see what happens (Figure 7). The quinine in tonic water is also fluorescent (Figure 8).


Other examples are some highlighter pen inks (Figure 9) and fluorescein, a fairly non-toxic dye that is used in medical diagnostics . Non-toxic, but can cause nausea and allergic reactions, so be careful with it. It was used to turn the Chicago River green for St. Patrick’s day in the 1960s, but a proprietary vegetable dye is used these days .
Chemiluminescence
Chemiluminescence is the production of light from chemical reaction. This includes the blue light from a natural gas burner or the bottom of a candle flame which typically comes from C2* or CH* radicals relaxing from an excited state to a ground state, and emitting blue photons in the process. The radicals are highly reactive molecules that exist only in the reaction region of the flame, and can be used to mark the flame region.
However chemiluminescence in general refers to light from chemical reactions without much heat. Party bracelets and Cyalume light sticks are examples. Two liquid reactants are separated by a fragile inner container, often glass. When broken, the reactants mix and a chemical reaction takes place, emitting photons of specific energy either immediately (as in fluorescence) or after a delay (as in phosphorescence). The color depends on exactly which chemicals are used . Note that these compounds are mildly toxic and certainly irritating to skin and eyes, and so should be handled with proper protective equipment (goggles, gloves) in a controlled environment. If you remove them from their commercial packaging they should be disposed of as hazardous waste; they should not be poured down the drain.
Another example of chemiluminescence is airglow , as shown in Figure 10. Molecules of OH, sodium and oxygen in the high (90 km) atmosphere give off characteristic photons when excited by UV light from the sun.

Due to the highly sensitive camera this photograph also showcases a mysterious phenomenon called airglow. The night sky is ablaze with deep red and eerie green hues, caused by the faint glow of Earth’s atmosphere. Because of airglow, no observatory site on Earth could ever be absolutely, completely dark — although ESO’s do come pretty close.
This image was taken by talented astronomer and photographer Yuri Beletsky, a member of the 2016 ESO Fulldome Expedition team. Y. Beletsky (LCO)/ESO, CC BY 4.0 , via Wikimedia Commons
Bioluminescence
When chemiluminescence is the product of a biochemical reaction it becomes bioluminescence. Generally a light-emitting compound, luciferin, is oxidized via catalysis by an enzyme, luciferinase . Ocean waves, ship, submarine and even human swimmers’ wakes can be visualized by the bioluminescence of Noctiluca scintillans or Lingulodinium polyedra , a widespread type of algae or plankton that glows or sparkles when disturbed, causing a phenomenon called mareel , as shown in Figure 11.
[videopress njsxGYkZ]
Figure 11 Bioluminescent algae (Lingulodinium polyedra) bloom in San Diego. April 27 2020, filmed by Dr. Tim Fallon using a Sony A7S camera .
Electroluminescence
Electroluminescence is when a material emits photons in response to excited states caused by electricity or an EM field. It is a relatively old technology, being used in night lights and instrument panels from the 1960s up to OLEDs today. It also includes sodium vapor and mercury vapor lights used as street lights. It occurs inside a fluorescent light when UV photons are produced by electrically excited mercury vapor atoms which then cause visible fluorescence in the phosphor coating the inside surface of the tube. Neon lights and LEDs can also be considered a type of electroluminescence. Its use in flow visualization has been primarily as a lighting source. One unusual application is the electric pickle. When a pickle is energized by 110V AC the sodium atoms in the pickle brine are stimulated to emit a somewhat sickly yellow light, as shown in Figure 12, plus a ferocious stench as the pickle burns.

I must say DO NOT ATTEMPT THIS AT HOME.
One final type of electroluminance to mention is the laser. My dad, Abraham Hertzberg, was one of the first developers of the gas dynamic laser . All lasers work by creating a population inversion, with more molecules in an excited state than a ground state; then the lasing medium can give off more photons than it absorbs. Most lasers in common use today are have solid state lasing media, but a gas dynamic laser uses a shock wave to produce a population inversion in a gas. It was a very high-power type of laser at the time, in the 1960s.
References

